Copolyester scores 10 out of 10 in every category
This post was initially going to enlighten you about what exactly copolyester is and where it comes from. However, while trying to research this matter I came across several matters of greater importance, mainly the fact that companies are actively hiding the exact information I was searching for.
Copolyester seems to be Nalgene’s upcoming favorite material. As showcased in these “nutrition facts,” Nalgene provides ample information highlighting the great qualities of copolyester. However, it fails to provide information about its production process or the materials used to produce it. Even, Nalgene Choice (www.nalgenechoice.com), a website designed specifically for consumer awareness of materials, was extremely unhelpful.
Nalgene Choice did not have any supplement information to Nalgene’s original website, and in fact seemed to be similar in nature to “green washing.” Because of this inadequacy of information online, I called Nalgene’s customer service hotline (1-800-625-4327) in attempts to find answers to my questions:
- Does Nalgene synthesize their own copolyester?
- If yes, please describe the production process in as much detail as possible. What raw materials are used? How are they combined to create copolyester? What byproducts are produced during the process?
- If no, what supplier does Nalgene purchase copolyester from? How does Nalgene manufacture the Everyday bottle once it receives copolyester? Are additional materials mixed with the copolyester, such as coloring dyes?
After speaking to several individuals I was connected to the Technical Support division where I talked with a surprisingly helpful worker. She informed me that Nalgene does not manufacture their own copolyester. Instead, they purchase Tritan copolyester in the form of resin pellets from Eastman, a company that manufactures chemicals, fibers and plastics used in a variety of products worldwide. Similarly to my efforts with Nalgene, I struggled to uncover relevant manufacturing information from Eastman’s website. I hit another dead end when I called their Corporate Headquarters (1 (423) 229-2000). I was connected to their “Education Initiatives,” but ended up reaching a voicemail. Equipped with my previous experience of failed returned calls, I decided I needed to contact them in a different way. I filled out the following product request form:
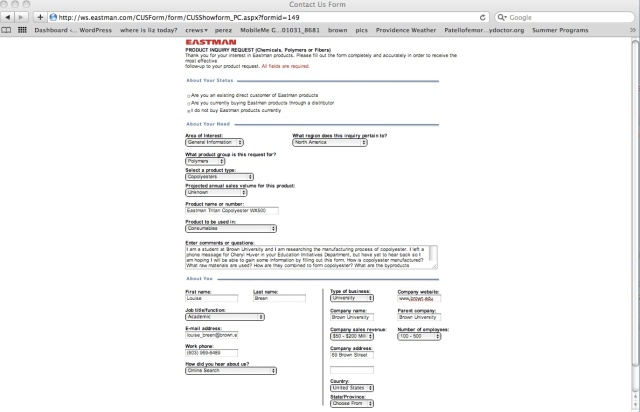
product request form
I’ll admit I stretched the truth a bit on the form by implying that I was an academic employed by Brown University, but the form required all fields to be completed. I received a less-than-enthusiastic response in the form of the following email exchange:

Email 1: this first email appeared to be an automated reply. I had already searched
http://www.eastman.com thoroughly so I replied with the email below in attempts to get more information.

Email 2: I thought this was a pretty polite email and I was expecting a helpful response answering my questions or directing me on the path to find answers. However, this was not the case.
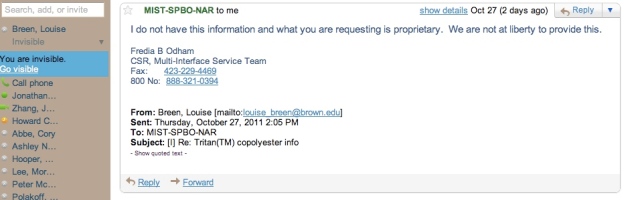
Email 3: Shutdown

Email 4: Although I asked for help again, I received no reply in response to this email.
I was accused of propriety! This was not anything I had planned on encountering during my research process and it seemed that it would be impossible for me to learn any information about the manufacturing process of copolyester. To top this off, the woman I spoke with at Nalgene told me that she also could not tell me the details of Nalgene’s manufacturing process because of legal reasons. At this point my panic began to rise rapidly. How was I ever supposed to trace the environmental impact of a Nalgene without being legally able to obtain any information about its composition or manufacturing process? Why would they not share this information with me? I told them I was a student doing a research for an environmental science project; I was not going to steal their production method and start up my own factory. I was only curious.

In addition to offering factory tours, Ben and Jerry's provides an extensive explanation of their manufacturing process all the way from "cow" to "cone." Each of the ice cream scoops in the above screen shot represent one step in the process, such as cows at family farms, a factory overview that includes video footage of their factory in action, the pint filler, and quality assurance. Eastman and Nalgene offer nothing like this online or elsewhere.
This raises an important question of what these companies are trying to hide. A great proportion of commonly used products have simple manufacturing processes that are freely shared or that we are already aware of. For example, as a child I visited the Ben and Jerry’s factory in Vermont, where I received a full tour that detailed where their ingredients came from and allowed us to observe the production and packaging processes. When I called Nalgene to ask where their factories were located, as well as if they offered factory tours, I spoke to several individuals who were clueless about the issue before being connected to a voicemail again. Nalgene and Eastman do not offer anywhere near the information Ben and Jerry’s and some other companies do their consumers. Why not? In my opinion, because Ben and Jerry’s is a good company that uses quality ingredients from local farmers in a safe, efficient and clean environment to produce a well-loved product. Showing customers the production process does not detract from the enjoyment one gains from eating a dish of Cherry Garcia; rather it enhances it.
This could not be farther from the truth of the copolyester in my Nalgene. I think that the manufacturing process of copolyester and Nalgenes is not necessarily a pretty process. What are these companies trying to hide? Eastman has tons of what seems to be propoganda avidly supporting the use of Tritan copolyester, but it refuses to share how they make it.
The propoganda of Tritan Copolyester:

Eastman tries to seem like it is a conscious company.

Eastman believes Tritan copolyester is perfect!

Using Tritan copolyester will save iguanas!

Eastman Tritan copolyester is the best!

Sure, Tritan copolyester is a winner, but where exactly is the Tritan copolyester in this advertisement? Is it used to produce the helmet, the water bottle or something else?

This reminds me of the "nutrition facts" created about the Everyday Nalgene. Similarly, the product being judged received 100% ratings.
How can there be so much of this support of the use of Tritan copolyester without any actual information about the product? My frustration was rising rapidly as I uncovered more and more pretty pictures and surface information about the benefits of Tritan copolyester, yet could not find a single bit of information about the manufacturing process or even the raw materials used to produce it. For example, the only relevant information from a press release regarding the creation of Tritan was, “Tritan™ is a result of unique chemistry based on a new commercial monomer.” Things initially looked hopeful when I came across the “Tritan Copolyester EX401” data sheet. It included many sections, such as hazards identification, compositions/information on ingredients, first aid measures, exposure controls, physical and chemical properties, toxicological information, ecological information, disposal considerations and transportation information. I’ve highlighted the data from the most important sections below:
1. Composition/information on ingredients
| Chemical name |
Concentration |
Additional Identification |
Notes |
| copolyester |
>90% |
proprietary |
|
| modifers/additives |
<10% |
proprietary |
|
*All concentrations are percent by weight unless ingredient is a gas. Gas concentrations are in percent by volume.
This section of the report emphasizes my already found knowledge that Eastman is simply not going to tell me what copolyester is made of, because of proprietary.
2. Information on Toxicological effects

toxicity to humans

toxicity to humans
toxicity to humans
Why did I include this long list of information with repetitive results of “no data available?” To showcase the point that not only does Eastman not share information with its consumers, but their own knowledge of their own products is extremely limited. This immediately brought to mind an important theme in Rachel Carson’s Silent Spring and our class discussion of the book. Carson addressed our country’s widespread use of many pesticides, specifically DDT, despite a lack of knowledge regarding its effects on humans or our environment. She implored the American public to force industry and government to investigate all of the consequences of chemicals and make this information widespread knowledge in order for appropriate decisions about the proper use of these materials could be made.
This copolyester data reveals an eerily similar lack of knowledge and makes me wish there was another push to investigate all of the consequences of this material so that it can be used appropriately. It is unacceptable that there is “no data available” regarding every single toxicological effect of Tritan copolyester! Does it cause cancer? Eastman admits they have no idea! (or worse, they are refusing to share the actual information because it would negatively impact their business) Does it cause reproductive problems? Again, not a clue! In addition to this lack of knowledge regarding the effects of Tritan copolyester on humans, there is an absence of information about an even more relevant component of this report: its environmental effects. Does this product effect animals such as fish? “No data available.” Is copolyester biodegradable? Either Eastman does not know or it will not share.

Ecological and environmental toxicity
What does all this lack of information boil down to for this report? I’ve fallen into the river of failure. The obtainment of copolyester is going to remain a mystery for me, for you and for all of the world locked behind the Berlin Wall of the Eastman. Although the use of Tritan copolyester is highly recommended by both Nalgene and Eastman, information regarding this substance’s composition and production is hidden from the public for legal reasons, while its broader effects have not been thoroughly researched. Despite my admittance that I will be unable to teach you about the obtainment and manufacture of this product, I can confidently say that this does not mean that there is a lack of environmental impacts of this. For example, Eastman has 21 manufacturing sites spread globally in places such as Brazil, China, the United States and Estonia. These factories require large quantities of land and energy that thus eliminates natural ecosystems and contributes to global warming. Additionally, even though copolyester is produced synthetically, there must be natural materials that need to be obtained due to the property that matter cannot be created or destroyed. Although I cannot pinpoint the exact effects of these, that does not mean that they do not exist. As a matter of fact, Eastman has analyzed the environmental impacts of the material production of Tritan copolyester when used to produce sport water bottles (such as the Everyday Nalgene or Camelbak). The graphs below show the breakdown of impacts during different stages of a bottle’s life, including bottle material, bottle manufacture, coatings and decoration, and end of life. Focus on the left bar of each graph for now, as that represents Eastman Tritan copolyester, whereas the others represent stainless steel and aluminum based water bottles.


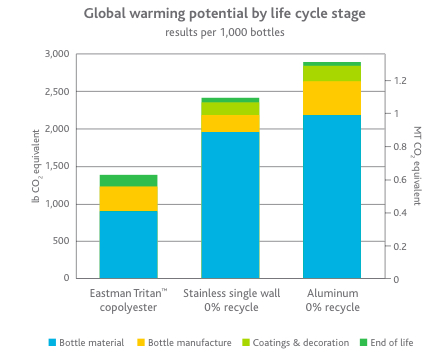 As you can see from these graphs, material production clearly contributes the largest environmental impacts in all three categories of energy input, global warming impacts and smog formation. These graphs show that on a large, worldwide scale, our use of Tritan copolyester water bottles contributes vast environmental impacts. It is time to address another cause of these impacts, represented by yellow in the graphs and addressed briefly at the beginning of this post: bottle manufacture.
As you can see from these graphs, material production clearly contributes the largest environmental impacts in all three categories of energy input, global warming impacts and smog formation. These graphs show that on a large, worldwide scale, our use of Tritan copolyester water bottles contributes vast environmental impacts. It is time to address another cause of these impacts, represented by yellow in the graphs and addressed briefly at the beginning of this post: bottle manufacture.